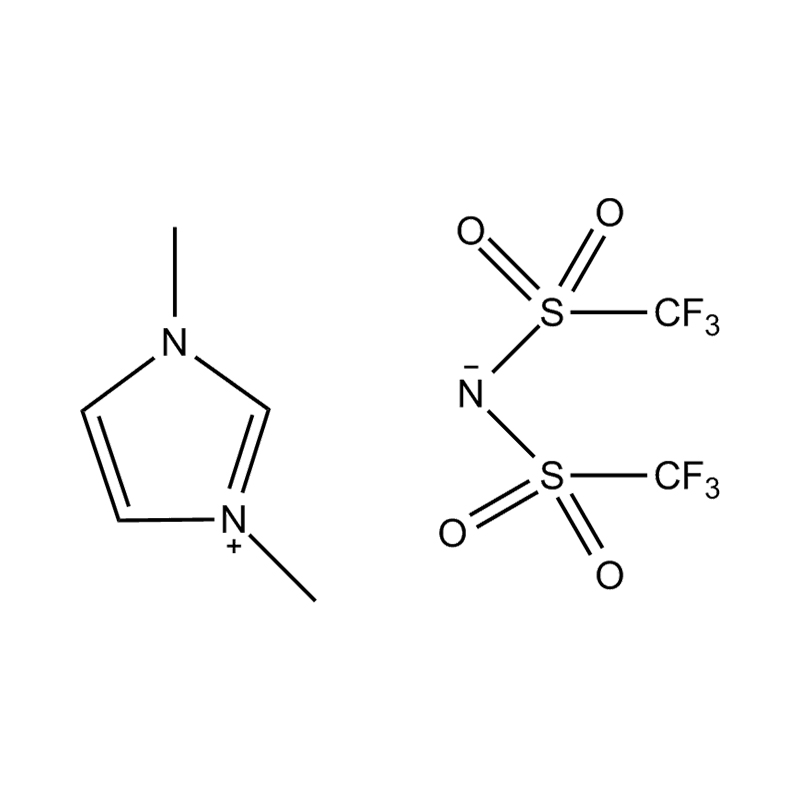
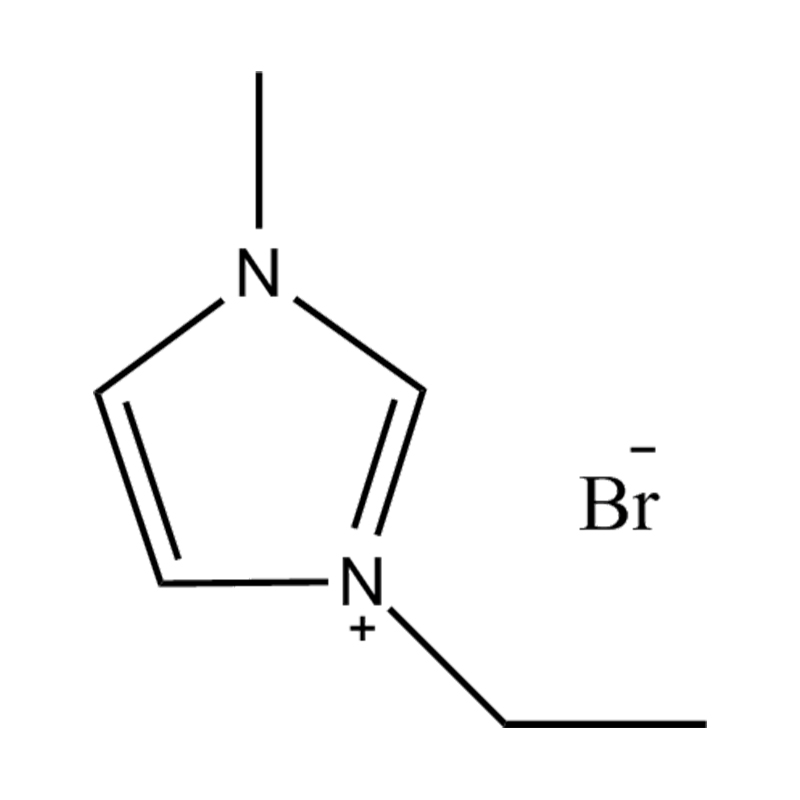
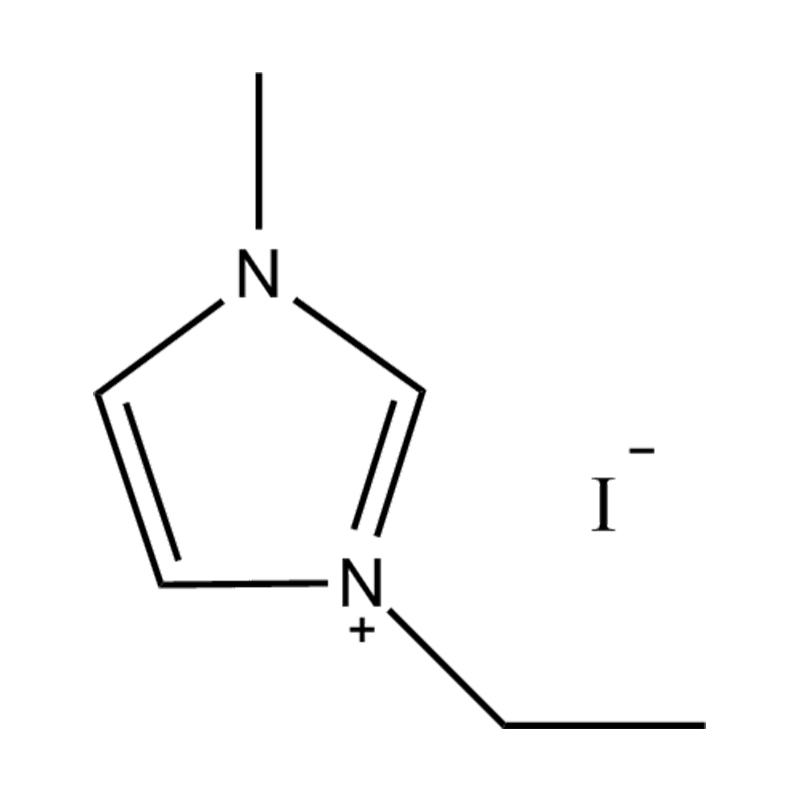
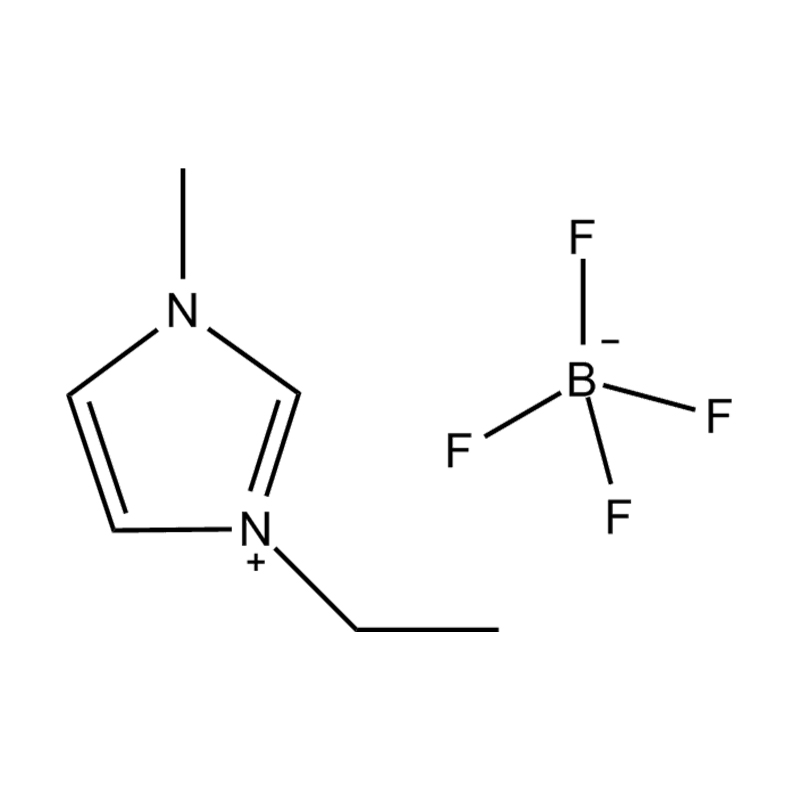
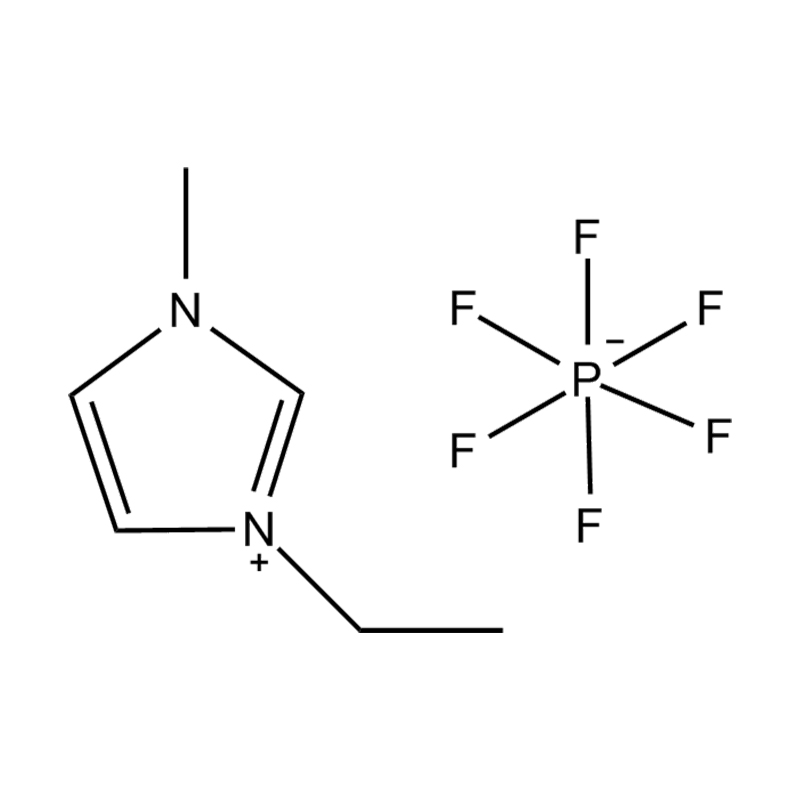
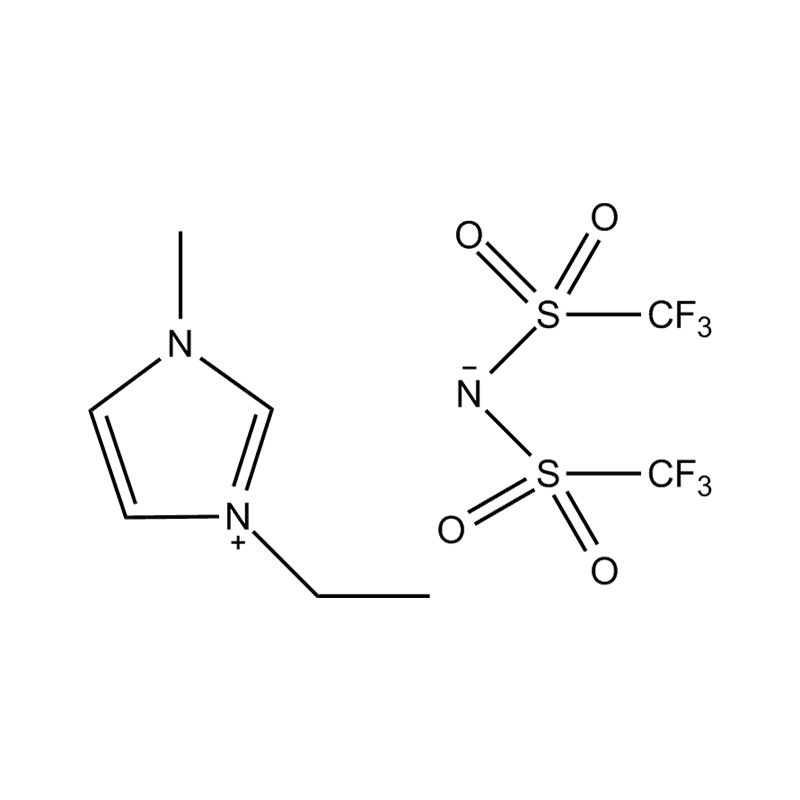
The cation of 1,3-dimethylimidazole bis(trifluoromethanesulfonyl)imide salt is 1,3-dimethylimidazole cation, which belongs to the type where the nitrogen atoms at positions 1 and 3 of the imidazole ring are respectively replaced by methyl groups. derivative. The anion is bis(trifluoromethanesulfonyl)imide, a larger, fluorinated, hydrophobic anion. The presence of NTf2 anions makes it hydrophobic and low-polar, and immiscible with many organic solvents, but has good compatibility with certain organic substances and non-polar compounds. Compared with many other ionic liquids, 1,3-dimethylimidazole bis(trifluoromethanesulfonyl)imide salt has a lower melting point and is usually liquid at room temperature. It can be used as a room temperature ionic liquid in many applications. It is easy to operate and does not require heating. It also has good electrical conductivity and relatively low viscosity, making it suitable for use in various electrochemical environments and is not miscible with water and other polar solvents.
1,3-Dimethylimidazole bis(trifluoromethanesulfonyl)imide salt is often used in the field of electrochemistry as an electrolyte material for batteries and supercapacitors. In addition, it is widely used as a catalyst and solvent in organic synthesis, and is especially suitable for separation technologies such as biphasic catalytic systems and liquid-liquid extraction.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体